Medicinal products that enter the healthcare market place must go through a rigorous clinical trial followed by careful post-marketing surveillance to ensure that no adverse outcome ensues that was missed during a limited clinical trial. The use of MPHO in therapy has not been subjected to the rigors of this type of regulatory process but rather has taken a different pathway based on the medical model of trial and error, with the expectation that the MPHO will perform or function in the recipient as it did in the donor. The focus, therefore, has been on validating processing and storage to demonstrate that they have not harmed the required national properties of the MPHO. Outcome data is often published but responses to scientific publications documenting adverse outcomes can take a long time before changes in practice are implemented. By combining the capture of such incidents, a more rapid response can be developed and tested for efficacy.
The basic elements of vigilance as mentioned in section 3, should include: adverse occurrence monitoring (for recipients and donors), product quality assurance (including processing controls and error management), and emerging threat assessment using epidemiologic and laboratory data (bioinformatics, repositories).
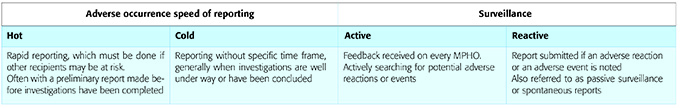
There are two main types of surveillance approaches to these issues
In the US a great example of success in vigilance has been in the recognition of the magnitude of healthcare-associated infections (HAI). Although this system of vigilance has been in place within local medical institutions for many years, the impact on patient safety and healthcare cost has been only recently realized. Patient safety and cost recovery data are only currently becoming available but indicate that huge cost savings can be made if proper vigilance systems are used as part of a total quality system. Only by starting to look at various processes within the healthcare setting can one become aware of the health burden and the impact on health economics. One must also be aware of the cost of each proposed action to improve safety, which could also result in the loss of donors and the subsequent potential loss of life for recipients.